New Genetic Tool Maps How Deadly Viruses Spread Around the World in Real Time

Tacio Philip Sansonovski/Shutterstock.com
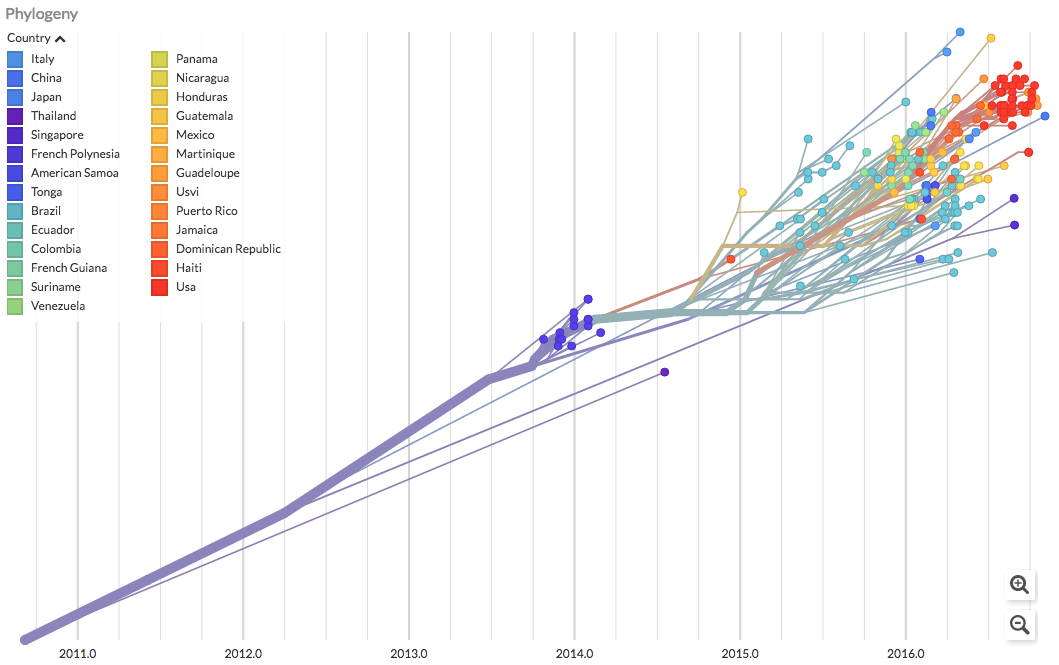
The real-time tracking tool maps how viruses spread, potentially identifying hot zones sooner.
We remain utterly unprepared to deal with epidemics at a global scale. Just think of Ebola, which killed more than 11,000 people, or Zika, which left thousands of newborns with malformed brains and has now become an endemic disease.
The good news is that new inventions are continually improving our odds of succeeding against the next epidemic. One such invention, a clever genetic tool that maps in real time how viruses spread, has just won the Open Science Prize, given for “unleashing the power of data to advance discovery and improve health.”
The researchers who developed it, Richard Neher of the University of Basel and Trevor Bedford of the Fred Hutchinson Cancer Research Center in Seattle, will each get $115,000 to make the tool accessible to scientists around the world and map disease-causing microbes beyond Ebola and Zika.
Viruses are scary because they can mutate and gain new powers much more quickly than any other kind of pathogen. At every transmission from one host to another, a virus’s DNA undergoes random tweaks to its small genome. In case of Ebola, for instance, one such transmission suspected to be from a wild animal to a 2-year-old toddler in Guinea gave the virus the mutation it needed to spread faster than it had before and kill thousands of people.
A Part-Time Project
When Neher and Bedford met at a conference, they hatched a plan to turn a virus’ modus operandi against it, by using it as a tracking tool. Researchers had begun taking genetic information of viruses during epidemics, but they didn’t have a single place to compare how and where different strains of viruses were mutating.
In their spare time, the two researchers built a prototype for influenza virus, because its genetic data is easily available and it is one of the most well-studied viruses. NextStrain was born. It just so happened as their tool became ready, the Ebola epidemic was peaking.
“On-the-ground researchers during the Ebola epidemic were collecting virus genetic sequences,” Neher told me, “but without putting them together, you don’t get the bigger picture.” Some of those researchers knew about the Neher-Bedford project and sent them their own viral DNA data. The duo used that data to expand its tool to include first Ebola and then Zika.
Though the tool came a little too late for turning data into action for the Ebola and Zika epidemics, Bedford described two scenarios where NextStrain could have helped. Looking back, we know Zika’s hot zone was in north-eastern Brazil. With a real-time tracking tool to map how the virus spread, this hot zone could have been discovered sooner. This would have allowed health officials to concentrate efforts there and perhaps limit the disease’s spread.
Another application could have been in Florida. Genetic analysis later showed more than one strain of the Zika virus was spreading in the region. That shows, for instance, it didn’t just spread between people in Florida, but that travelers continued bringing in new strains of the disease. So screening those coming in may have been more important than killing more mosquitoes.
This kind of small tweak in attacking an epidemic, especially when resources are limited, might have gone a long way in stopping the disease from spreading to as many as 76 countries and territories around the world.
To be sure, NextStrain can’t yet help in predicting outbreaks. Even though we can now observe viruses mutating, we don’t know which mutation will give the virus the qualities it needs to start an epidemic. Still, NextStrain is useful for tracking the spread of a disease once it has begun and blunting its assault.
Changing the Culture
Why didn’t such a seemingly simple and useful tool as NextStrain already exist?
“I don’t think there had been a hole in the scientific marketplace for this kind of tool until recently,” Bedford told me. That’s because it’s only recently scientists have been able to take miniature gene-sequencing machines such as Oxford Nanopore’s minION to the field to get data instantly. So genetically tracking an epidemic as it happens has only been possible since Ebola.
NextStrain faces another battle: getting more scientists to use it. That’s because scientific-journal articles, which are the currency of academic prestige, require that data not be shared before publication. The upshot is that, typically, researchers guard their data for months after they collect it. But in an epidemic, data need to be shared so health agencies can take action.
That culture may be changing, Neher told me.
“Researchers who worked during the Ebola and Zika epidemics understand that data sharing is imperative,” he said. “At the time, journals too put out explicit policies allowing pre-sharing of data for those diseases.”
Neher and Bedford now plan to expand NextStrain to include viruses such as Middle East Respiratory Syndrome and norovirus, and perhaps even bacteria, such as drug-resistant New Delhi metallo-beta-lactamase 1, which have much bigger genomes than viruses.
The Open Science Prize recognizes the tool’s potential. Expanding it to be globally useful will require a lot more money. The prize is funded by the National Institutes of Health, the Howard Hughes Medical Institute and the biomedical charity Wellcome.
When the prize money runs out in a couple of years, Neher said, “We’re not averse to a public-health agency taking over to keep running it.” The underlying software is open-source and thus free for anybody to copy and use. Neher has talked to officials at the World Health Organization and he says the agency doesn’t, as yet, have the kind of expertise needed to run NextStrain





